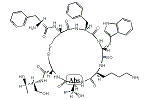
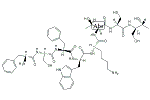
オクトレオチド
| 分子式: | C49H66N10O10S2 |
| その他の名称: | SMS-201-995、オクトレオチド、Octreotide、D-Phe-L-Cys(1)-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys(1)-N-[(1R,2R)-2-Hydroxy-1-(hydroxymethyl)propyl]-NH2、SMS-201995、D-Phe-Cys(1)-Phe-D-Trp-Lys-Thr-Cys(1)-N-[(1R,2R)-1-(Hydroxymethyl)-2-hydroxypropyl]-NH2 |
| 体系名: | D-Phe-L-Cys(1)-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys(1)-N-[(1R,2R)-2-ヒドロキシ-1-(ヒドロキシメチル)プロピル]-NH2、D-Phe-Cys(1)-Phe-D-Trp-Lys-Thr-Cys(1)-N-[(1R,2R)-1-(ヒドロキシメチル)-2-ヒドロキシプロピル]-NH2 |
D-Phe-L-Cys-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys-N-[(1R,2R)-2-ヒドロキシ-1-(ヒドロキシメチル)プロピル]-NH2
| 分子式: | C49H68N10O10S2 |
| その他の名称: | SMS-201-995【reduced】、Octreotide【reduced】、オクトレオチド【還元型】、D-Phe-L-Cys-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys-N-[(1R,2R)-2-Hydroxy-1-(hydroxymethyl)propyl]-NH2、(2R,3R)-2-[(D-Phe-L-Cys-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys-)Amino]-1,3-butanediol |
| 体系名: | D-Phe-L-Cys-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys-N-[(1R,2R)-2-ヒドロキシ-1-(ヒドロキシメチル)プロピル]-NH2、(2R,3R)-2-[(D-Phe-L-Cys-L-Phe-D-Trp-L-Lys-L-Thr-L-Cys-)アミノ]-1,3-ブタンジオール |
オクトレオチド
オクトレオチド
出典: フリー百科事典『ウィキペディア(Wikipedia)』 (2022/12/12 09:45 UTC 版)
 |
|
 |
|
| IUPAC命名法による物質名 | |
|---|---|
|
|
| 臨床データ | |
| 販売名 | Sandostatin |
| Drugs.com | monograph |
| 胎児危険度分類 | |
| 法的規制 | |
| 投与方法 | Subcutaneous, intramuscular, intravenous |
| 薬物動態データ | |
| 生物学的利用能 | 60% (IM), 100% (SC) |
| 血漿タンパク結合 | 40–65% |
| 代謝 | Hepatic |
| 半減期 | 1.7–1.9 hours |
| 排泄 | Urine (32%) |
| 識別 | |
| CAS番号 |
83150-76-9  79517-01-4 (acetate) 135467-16-2 (pamoate) |
| ATCコード | H01CB02 (WHO) |
| PubChem | CID: 448601 |
| IUPHAR/BPS | 2055 |
| DrugBank | DB00104en:Template:drugbankcite |
| ChemSpider | 395352 |
| UNII | RWM8CCW8GP  |
| KEGG | D00442 en:Template:keggcite |
| ChEMBL | CHEMBL1680en:Template:ebicite |
| 化学的データ | |
| 化学式 | C49H66N10O10S2 |
| 分子量 | 1019.24 g/mol |
|
|
|
|
オクトレオチド(Octreotide)はソマトスタチン模倣オクタペプチドである。ソマトスタチンよりも成長ホルモン、グルカゴン、インスリン阻害作用が強い。商品名サンドスタチン。
効能・効果
- (筋・皮)消化管ホルモン産生腫瘍(VIP産生腫瘍、カルチノイド症候群の特徴を示すカルチノイド腫瘍、ガストリン産生腫瘍)の諸症状の改善
- (筋・皮)先端巨大症・下垂体性巨人症(外科的処置、他剤による治療で効果が不十分な場合または施行が困難な場合)における成長ホルモン、ソマトメジンC分泌過剰状態および諸症状の改善
- (筋のみ)消化管神経内分泌腫瘍
- (皮のみ)進行・再発癌患者の緩和医療における消化管閉塞に伴う消化器症状の改善
腫瘍
オクトレオチドは成長ホルモン産生腫瘍(先端巨大症および巨人症)、甲状腺刺激ホルモン産生脳下垂体腫瘍、カルチノイド症候群による下痢および潮紅、血管作動性腸管ペプチド産生腫瘍(VIPオーマ)による下痢の治療に使われる。
食道静脈瘤出血
オクトレオチドはしばしば肝硬変で門脈圧が亢進している場合の食道静脈瘤からの急性出血の治療に静脈注射で用いられるが、効果は限定的であり生存率の向上には繋がらない[3]。
放射線標識化
オクトレオチドにキレート剤を結合して111Inを担持させた薬剤はソマトスタチン受容体を発現している神経内分泌腫瘍などの非侵襲的シンチグラフィに用いられる[4]。最近は11C[5]や68Gaを用いたポジトロン断層法(PET)も実施され、解像度や感度の改善に寄与している。
オクトレオチドは90Yや177Luなどの種々の放射性核種をキレートして切除不能の神経内分泌腫瘍の放射性核種標識ペプチド治療(Peptide receptor radionuclide therapy、PRRT)に応用されている[6]。
標的受容体
オクトレオチドはSRIF受容体とも呼ばれるソマトスタチン受容体に結合する。SRIF受容体は神経内分泌腫瘍(NET)等で特に過剰発現している。SRIF受容体には5種類(SSTR1〜SSTR5)が知られており、腫瘍のタイプ毎にどの種類が発現しているかが異なる。例えば胃腸膵管系神経内分泌腫瘍(GEP-NET)ではSSTR2の発現が多く、SSTR1とSSTR5は少ない[7]。
禁忌
オクトレオチドの小児・妊婦・授乳婦に対する影響は検討されていない。これらの患者には、危険便益分析で便益の方が高いと判断された場合にのみ使用すべきである[8][9]。
副作用
添付文書に記載されている重大な副作用は、アナフィラキシー(頻度不明)と徐脈(0.1%)である[1][2]。
10%以上の患者に、頭痛、甲状腺機能低下症、刺激伝導系変化、消化器系症状(腹痛、嘔気、嘔吐、下痢、便秘等)、胆石、インスリン分泌低下、高血糖症[10]、低血糖症、(一過性の)注射部位反応が発生する。また1%以上の患者に徐脈、瘙痒などの皮膚反応、ビリルビン上昇、甲状腺機能低下症、眩暈、呼吸困難が発生する。稀な副作用としては、アナフィラキシー、膵炎、肝炎が知られている[8][9]。関節リウマチとの関連を示唆した研究もある[11]。脱毛の報告もある[12]。ラットも用いた1998年の実験では、勃起不全が見られた[13]。
QT時間の延長が報告された事があるが、薬剤の反応か疾患によるものかは定かではない[8]。
| 識別子 | |
|---|---|
| 略号 | N/A |
| OPM superfamily | 167 |
| OPM protein | 1soc |
相互作用
オクトレオチドは消化管でのシクロスポリン吸収を減少させるので使用時はシクロスポリンの投与量増加が必要となることがある[14]。
糖尿病患者はインスリンや経口血糖降下薬の必要量が変化(増加または減少)し得る。
ブロモクリプチンの生物学的利用能は増大する[9]。ブロモクリプチンは抗パーキンソン病薬であるが、先端巨大症の治療にも用いられる。
内分泌への影響
オクトレオチドのソマトスタチン様作用によって次の様な影響がある。
- 種々のホルモン、ガストリン、コレシストキニン、グルカゴン、成長ホルモン、インスリン、セクレチン、膵臓ポリペプチド、甲状腺刺激ホルモン、血管作動性腸管ペプチドなどの分泌低下
- 腸および膵臓からの分泌液の減少
- 消化管運動低下と胆嚢収縮抑制
- 脳下垂体前葉ホルモンの作用阻害
- 血管収縮
- 静脈瘤出血部位の血管圧低下
オクトレオチドは鎮痛剤様の作用を持ち、μ-オピオイド受容体の部分作動薬である[15][16]。
薬物動態
筋肉内注射
筋注時、オクトレオチドの血中濃度は25〜34日で最高血中濃度Cmaxに達し、その前後でCmaxの80%以上であった期間は17〜19日間であった[1]。
皮下注時、オクトレオチドは速やかに完全に吸収され、およそ0.6時間後にCmaxに達した。その後の血中半減期 t1/2は平均1.78時間であった[2]。
点滴静注した際には、オクトレオチドの消失は2相性を示し、それぞれの半減期は10分と90分であった[8][9]。
適応外使用等
オクトレオチドは重篤な反復性の下痢の治療に適応外使用される。毒物学の分野で、スルホニルウレア過量投与後の遷延性低血糖の治療に用いられる。幼児の膵島細胞症におけるインスリン分泌過剰に対する効果は一定していない。群発頭痛に対する治療薬(発作解除)としての臨床試験がいくつか実施され、オクトレオチドの皮下注が偽薬より有効性が高いことが示された[17]。
オクトレオチドは肥満の治療薬としても実験的に使用され、視床下部の空腹中枢または満腹中枢に以上がある場合は特に有効であるとされた[18]。視床下部の弓状核に始まる回路は外側視床下部(LH)および視床下部腹内側部(VMH)に終わり、それぞれ脳の摂食および満腹中枢である[19][20]。VMHはしばしば急性リンパ性白血病(ALL)の治療や手術、後頭蓋窩腫瘍の放射線治療で障害を受ける[18]。VMHが傷害されると末梢のエネルギーバランスシグナルに応答しなくなり、遠心性交感神経活動が低下して倦怠感を感じると共にエネルギー消費が減少し、迷走神経が亢進してインスリン分泌が増加し脂肪生成が増加する[21]。VMH機能不全はカロリーの過剰摂取を促進すると共にカロリー消費量を減少させ、継続性で難治性の体重増加をきたす。この症状に対してカロリー制限やアドレナリン系、セロトニン系薬剤を用いた治療が成功した事は過去にほとんどない[18]。オクトレオチドは過度のインスリン分泌を抑え、活動性を増加させ、過剰の脂肪蓄積を抑制する。ALLまたは脳腫瘍治療後で他に視床下部機能不全の診断根拠を有する難治性体重増加の小児患者18名を対象とした小規模治験では、オクトレオチドは偽薬よりもBMIを低下させ、経口ブドウ糖負荷試験でのインスリン分泌を抑制し、親の報告した身体活動性およびQOLを向上させた[18]。視床下部腫瘍がない肥満患者に長時間型オクトレオチド製剤を投与した別の臨床試験では、偽薬よりも体重およびBMIが減少し、後追い解析では、高用量の患者またはコホートの中央値よりインスリン分泌の多いコーカソイドの患者で効果が大きかった。QOLスコア、体脂肪、血中レプチン濃度、ベックうつ病特性尺度、主栄養素摂取量では有意差が見られなかったが、オクトレオチド投与群では血糖負荷試験後の血糖値が偽薬群よりも高かった[22]。
オクトレオチドは慢性膵炎の疼痛に対する効果も検討されている[23]。
ミドドリンとの併用で肝腎症候群による末梢血管拡張に使用できる。この疾患では全身の血管抵抗が増大しているため、薬剤でシャントを減少させて腎臓の灌流を改善し、肝臓移植で根本治療するまでの時間稼ぎができる[25]。同様に、オクトレオチドは治療抵抗性低血圧にも使用できる[26]。
乳糜胸に対する治療成功例が症例報告される一方[27][28]臨床研究では有効性を示す事ができなかった[29]。
特発性頭蓋内圧亢進症の治療に有効であったとの小規模臨床試験が報告されている[30][31]。
出典
- ^ a b c “サンドスタチンLAR筋注用10mg/サンドスタチンLAR筋注用20mg/サンドスタチンLAR筋注用30mg 添付文書” (2015年3月). 2015年9月15日閲覧。
- ^ a b c “サンドスタチン皮下注用 50µg/サンドスタチン皮下注用 100µg 添付文書” (2015年3月). 2015年9月15日閲覧。
- ^ Gøtzsche PC, Hróbjartsson A (2008). “Somatostatin analogues for acute bleeding oesophageal varices”. Cochrane Database Syst Rev (3): CD000193. doi:10.1002/14651858.CD000193.pub3. PMID 18677774.
- ^ Medscape: Octreoscan review
- ^ Joshua Chin, Matthew Vesnaver, Vadim Bernard-Gauthier, Erin Saucke-Lacelle, Björn Wängler, Carmen Wängler, Ralf Schirrmacher. Amino Acids: Direct one-step labeling of cysteine residues on peptides with 11C-methyl triflate for the synthesis of PET radiopharmaceuticals. Amino Acids. 2013 Aug 7. PMID 23921782
- ^ “カルチノイド腫瘍に対する放射性ソマトスタチンアナログ療法”. Nature Japan (2010年8月1日). doi:10.1038/nrendo.2010.94. 2015年9月15日閲覧。
- ^ Prasad V, Fetscher S, Baum RP. (2007). “Changing role of somatostatin receptor targeted drugs in NET: Nuclear Medicine's view.”. J Pharm Pharm Sci. 10 (2): 321s-337s. PMID 17718935.
- ^ a b c d Haberfeld, H, ed (2009) (German). Austria-Codex (2009/2010 ed.). Vienna: Österreichischer Apothekerverlag. ISBN 3-85200-196-X
- ^ a b c d Dinnendahl, V, Fricke, U, ed (2010) (German). Arzneistoff-Profile. 8 (23 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. ISBN 978-3-7741-9846-3
- ^ Hovind P, Simonsen L, Bülow J (March 2010). “Decreased leg glucose uptake during exercise contributes to the hyperglycaemic effect of octreotide”. Clin Physiol Funct Imaging 30 (2): 141–5. doi:10.1111/j.1475-097X.2009.00917.x. PMID 20132129.
- ^ Saif MW (July 2011). “Rheumatoid arthritis associated with the use of Sandostatin LAR depot in a patient with pancreatic neuroendocrine tumor. An association or a coincidence? The first case report”. JOP 12 (4): 425–8. PMID 21737909. Lay summary – eHealthMe.com.
- ^ van der Lely AJ, de Herder WW, Lamberts SW (November 1997). “A risk-benefit assessment of octreotide in the treatment of acromegaly”. Drug Saf 17 (5): 317–24. doi:10.2165/00002018-199717050-00004. PMID 9391775.
- ^ Kapicioglu S, Mollamehmetoglu M, Kutlu N, Can G, Ozgur GK (January 1998). “Inhibition of penile erection in rats by a long-acting somatostatin analogue, octreotide (SMS 201-995)”. Br J Urol 81 (1): 142–5. doi:10.1046/j.1464-410x.1998.00520.x. PMID 9467491.
- ^ Klopp, T, ed (2010) (German). Arzneimittel-Interaktionen (2010/2011 ed.). Arbeitsgemeinschaft für Pharmazeutische Information. ISBN 978-3-85200-207-1
- ^ Maurer R, Gaehwiler BH, Buescher HH, Hill RC, Roemer D. Opiate antagonistic properties of an octapeptide somatostatin analog. Proceedings of the National Academy of Sciences USA. 1982 Aug;79(15):4815-7. PMID 6126877
- ^ Allen MP, Blake JF, Bryce DK, Haggan ME, Liras S, McLean S, Segelstein BE. Design, synthesis and biological evaluation of 3-amino-3-phenylpropionamide derivatives as novel mu opioid receptor ligands. Bioorganic and Medicinal Chemistry Letters. 2000 Mar 20;10(6):523-6. PMID 10741545
- ^ Subcutaneous octreotide in cluster headache: randomized placebo-controlled double-blind crossover study.
- ^ a b c d Lustig RH, Hinds PS, Ringwald-Smith K, Christensen RK, Kaste SC, Schreiber RE, Rai SN, Lensing SY, Wu S, Xiong X (June 2003). “Octreotide therapy of pediatric hypothalamic obesity: a double-blind, placebo-controlled trial”. J. Clin. Endocrinol. Metab. 88 (6): 2586–92. doi:10.1210/jc.2002-030003. PMID 12788859.
- ^ Flier JS (2004). “Obesity wars: Molecular progress confronts an expanding epidemic”. Cell 116 (2): 337–50. doi:10.1016/S0092-8674(03)01081-X. PMID 14744442.
- ^ Boulpaep, Emile L.; Boron, Walter F. (2003). Medical physiologya: A cellular and molecular approach. Philadelphia: Saunders. p. 1227. ISBN 0-7216-3256-4
- ^ Lustig RH (2011). “Hypothalamic obesity after craniopharyngioma: mechanisms, diagnosis, and treatment”. Front Endocrinol (Lausanne) 2: 60. doi:10.3389/fendo.2011.00060. PMC 3356006. PMID 22654817.
- ^ Lustig RH, Greenway F, Velasquez-Mieyer P, Heimburger D, Schumacher D, Smith D, Smith W, Soler N, Warsi G, Berg W, Maloney J, Benedetto J, Zhu W, Hohneker J (February 2006). “A multicenter, randomized, double-blind, placebo-controlled, dose-finding trial of a long-acting formulation of octreotide in promoting weight loss in obese adults with insulin hypersecretion”. Int J Obes (Lond) 30 (2): 331–41. doi:10.1038/sj.ijo.0803074. PMC 1540404. PMID 16158082.
- ^ Uhl W, Anghelacopoulos SE, Friess H, Büchler MW (1999). “The role of octreotide and somatostatin in acute and chronic pancreatitis”. Digestion 60 Suppl 2: 23–31. doi:10.1159/000051477. PMID 10207228.
- ^ Shima Y, Ohtsu A, Shirao K, Sasaki Y (May 2008). “Clinical efficacy and safety of octreotide (SMS201-995) in terminally ill Japanese cancer patients with malignant bowel obstruction”. Jpn. J. Clin. Oncol. 38 (5): 354–9. doi:10.1093/jjco/hyn035. PMID 18490369.
- ^ Skagen C, Einstein M, Lucey MR, Said A (Feb 2009). “Combination Treatment With Octreotide, Midodrine, and Albumin Improves Survival in Patients With Type 1 and Type 2 Hepatorenal Syndrome.”. J Clin Gastroenterol. 43 (7): 680–5. doi:10.1097/MCG.0b013e318188947c. PMID 19238094.
- ^ Patient.info (Feb 2013). Hypotension.
- ^ Kilic D, Sahin E, Gulcan O, Bolat B, Turkoz R, Hatipoglu A (2005). “Octreotide for treating chylothorax after cardiac surgery”. Tex Heart Inst J 32 (3): 437–9. PMC 1336729. PMID 16392238.
- ^ Siu SL, Lam DS (2006). “Spontaneous neonatal chylothorax treated with octreotide”. J Paediatr Child Health 42 (1-2): 65–7. doi:10.1111/j.1440-1754.2006.00788.x. PMID 16487393.
- ^ Chan EH, Russell JL, Williams WG, Van Arsdell GS, Coles JG, McCrindle BW (November 2005). “Postoperative chylothorax after cardiothoracic surgery in children”. Ann. Thorac. Surg. 80 (5): 1864–70. doi:10.1016/j.athoracsur.2005.04.048. PMID 16242470.
- ^ Greek Researchers Investigate Octreotide Hypertension Research Foundation, accessed 2011-01-02
- ^ Panagopoulos GN, Deftereos SN, Tagaris GA, Gryllia M, Kounadi T, Karamani O, Panagiotidis D, Koutiola-Pappa E, Karageorgiou CE, Piadites G (2007). “Octreotide: a therapeutic option for idiopathic intracranial hypertension”. Neurol Neurophysiol Neurosci: 1. PMID 17700925.
関連項目
- オクトレオテート - 類縁ペプチド
固有名詞の分類
- オクトレオチドのページへのリンク