Abstract
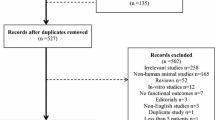
Traumatic brain injury is a major problem worldwide. Our objective is to synthesize available evidence in the literature concerning the effectiveness of neuroprotective drugs (cerebrolysin, citicoline, and piracetam) on Glasgow outcome score (GOS), cognitive performance, and survival in traumatic brain injury patients. Comprehensive search of electronic databases, search engines, and conferences proceedings; hand search journals; searching reference lists of relevant articles, theses, and local publications; and contact of authors for incomplete data were performed. Studies included patients in all age groups regardless of severity of trauma. There was no publication date restriction. Two reviewers independently extracted data from each study. Fixed effect or random effects model selection depends on results of statistical tests for heterogeneity. The literature search yielded 13 studies. Patients treated with cerebrolysin (n = 112) had favorable GOS three times more than controls (OR 3.019; 95 % CI 1.76 to 5.16; p = 0.003*). The odds of cognition improvement in the treatment group was 3.4 times more than controls (OR 3.4; 95 % CI 1.82 to 5.21; p < 0.001*). Survival of cerebrolysin-treated patients did not differ from controls (103 patients; OR = 2.81; 95 % CI 0.905 to 8.76). Citicoline did not improve GOS (1355 patients; OR 0.96; 95 % CI 0.830 to 1.129; p = 0.676), cognitive performance (4 studies; 1291 patients; OR 1.35; 95 % CI 0.58 to 3.16; p = 0.478), and survival (1037 patients; OR = 1.38; 95 % CI 0.855 to 2.239). One study showed a positive effect of piracetam on cognition. Further research with high validity is needed to reach a solid conclusion about the use of neuroprotective drugs in cases of brain injury.











Similar content being viewed by others
References
A’lvarez XA, Sampedro C et al (2008) Reduction in qEEG slowing over 1 year and after treatment with Cerebrolysin in patients with moderate-Severe traumatic brain injury. J Neural Transm 115:683–692
A’lvarez XA, Sampedro C, Pe’rez P, Laredo M, Couceiro V, Herna’ndez A et al (2003) Positive effects of Cerebrolysin on electroencephalogram slowing, cognition and clinical outcome in patients with postacute traumatic brain injury: an exploratory study. Int Clin Psychopharmacol 18:1–8
A’lvarez XA, Sampedro C, Pe’rez P, Laredo M et al (2003) Effect of Cerebrolysin on Quantitative EEG activity in elderly controls and in patients with brain damage. Eur Neuropsychopharmacol 13
Aboukhabar H, Abouelela A, Shebl M (2011) Impact of administration of the neuroprotective drug cerebrolysin alone and with vitamin e on outcome of patients with severe traumatic brain injury. AJAIC 13:1–11
Adibhatla RM (2013) Citicoline in stroke and TBI clinical trials. Nat Rev Neurol:1–2
Alvarez XA, Sampedro C, Pérez P (2003) Positive effects of cerebrolysin on electroencephalogram slowing, cognition and clinical outcome in patients with postacute traumatic brain injury: an exploratory study. Int Clin Psychopharmacol 18:271–278
American Association of neurological surgeon (AANS), Congress of Neurological Surgeons (CNS), AANS/CNS Joint Section on Neurotrauma and Critical Care (2007) Guidelines for the management of severe traumatic brain injury. J Neurotrauma 24:1–116
Aniruddha TJ, Pillai S, Devi BI, Sampath S, Chandramouli BA (2009) Role of citicoline in the management of mild head injury. Indian J Neurotrauma 6:49–52
Asghari M, Meshkini A, Salehpoor F, Aghazadeh, Shakeri M, Shokohi G (2014) Investigation of the effect of cerebrolysin on patients with head trauma and diffuse axonal injury. Int J Curr Res Acad Rev 2:1–8
Beattie MS, Ferguson AR, Bresnahan JC (2010) AMPA-receptor trafficking and injury-induced cell death. Eur J Neurosci 32:290–297
Borenstein M, Hedges LV, Higgins JP, Rothstein RH (2009) Converting among effect sizes. In: Introduction to meta-analysis. Wiley, London, pp 45–49
Borenstein M, Hedges LV, Higgins JP, Rothstein RH (2009) Heterogeneity. In: Introduction to meta-analysis. Wiley, London, pp 105–106
Borenstein M, Hedges LV, Higgins JP, Rothstein RH (2009) Converting among effect sizes. In: Introduction to Meta-Analysis. Wiley, pp 1–413
Borenstein M, Hedges LV, Higgins JP, Rothstein RH (2009) Fixed-effect versus random-effects models. In: Introduction to Meta-Analysis. Wiley
Borenstein M, Hedges LV, Higgins JP, Rothstein RH (2009) Publication bias. In: Introduction to Meta-Analysis. Wiley, pp 1–413
Chen CC, Tsaia SC, Chen XX, Cho DY (2013) Cerebrolysin enhances cognitive recovery of mild traumatic brain injury patients: double-blind, placebo-controlled, randomized study. Br J Neurosurg:1–5
Dempsey RJ, Raghavendra Rao VL (2003) Cytidinediphosphocholine treatment to decrease traumatic brain injury-induced hippocampal neuronal death, cortical contusion volume, and neurological dysfunction in rats. J Neurosurg 98:867–873
Donkin JJ, Vink R (2010) Mechanisms of cerebral edema in traumatic brain injury: therapeutic developments. Curr Opin Neurol 23:293–299
El Gindi S, Mahdy M, Abdel Azeem A (2001) Traumatic brain injuries in developing countries. Revista Espagnola de Neuropsicologia 3:3–11
Guyatt GH, Oxman AD, Vist GE et al (2008) GRADE Working Group: GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336:924–926
Higgins JPT, Green S (editors) (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration
Iznak FA, Iznak VE, Zavadenko NN, Guzilova SL (2008) Neurophysiological indices of CNS plasticity in the course of treatment of head trauma in adolescents. Hum Physiol 34:672–677
Jain KK (2011) The handbook of neuroprotection (neuroprotection in traumatic brain injury). Springer Science & Business Media:271–254
Levin HS (1991) Treatment of postconcussional symptoms with CDP-choline. J Neurol Sci 103:1–4
Lozano R (1991) CDP-choline in the treatment of cranio-encephalic traumata. J Neurol Sci 103, Supplement:43–47. doi:10.1016/0022-510X(91)90008-U
Maldonado V, Perez JBC, Escario JAE (1991) Effects of CDP-choline on the recovery of patients with head injury. J Neurol Sci 103:1–4
Meierhans R, Béchir M, Ludwig S (2010) Brain metabolism is significantly impaired at blood glucose below 6 mM and brain glucose below 1 mM in patients with severe traumatic brain injury. Crit Care 14:1–13
Mondello S, Papa L, Buki A (2011) Neuronal and glial markers are differently associated with computed tomography findings and outcome in patients with severe traumatic brain injury: a case control study. Crit Care J 15:R156
Onose G, Mureşanu DF, Ciurea AV, Chendreanu CD, Mihaescu AS et al (2009) Neuroprotective and consequent neurorehabilitative clinical outcomes, in patients treated with the pleiotropic drug cerebrolysin. J Med Life 2:350–360
Petzold A, Tisdall MM, Girbes AR (2011) In vivo monitoring of neuronal loss in traumatic brain injury: a microdialysis study. Brain 134:464–483
Sample PL, Johns N, Gabella B, Langlois J (2004) Can traumatic brain injury surveillance systems be used to link individuals with TBI to services? Brain 18:1177–1189
Secades JJ (2011) Citicoline: pharmacological and clinical review, 2010 update. Rev Neurol 52:1–63
Stahel PF, Smith WR, Moore EE (2008) Hypoxia and hypotension, the ‘lethal duo’ in traumatic brain injury: implications for prehospital care. Intensive Care Med 34:402–404
Teasdale G, Jennett B (1974) Assessment of coma and impaired sonciousness. A practical scale. Lancet 2:81–84
Vilalta A, Sahuquillo J, Merino MA (2011) Normobaric hyperoxia in traumatic brain injury: does brain metabolic state influence the response to hyperoxic challenge? J Neurotrauma 28:1139–1148
Wai S Poon The CAPTAIN Trial: Cerebrolysin Asian Pacific Trial in Acute Brain Injury and Neurorecovery. ClinicalTrialsgov Identifier:NCT01606111
Walcott BP, Kahle KT, Simard JM (2012) Novel treatment targets for cerebral edema. Neurotherapeutics 9:65–72
Wong GKC, Zhu XL, Poon WS (2005) Beneficial effect of cerebrolysin on moderate and severe head injury patients: result of a cohort study. Acta Neurochir:59–60
Xu Y, McArthur DL, Alger JR (2010) Early nonischemic oxidative metabolic dysfunction leads to chronic brain atrophy in traumatic brain injury. J Cereb Blood Flow Metab 30:883–894
Zafonte RD, Bagiella E, Ansel BM, Novack TA, Friedewald WT, Hesdorffer DC, Timmons SD (2012) Effect of Citicoline on functional and cognitive status among patients with traumatic brain injury citicoline brain injury treatment trial (COBRIT). JAMA 308:1–8
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Funding agencies had no role in the design or conduct of the study, including study identification, collection, management, analysis and interpretation of the data, or preparation, review of the final manuscript for publication. No role for institutional financial support.
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 29 kb)
Rights and permissions
About this article
Cite this article
El Sayed, I., Zaki, A., Fayed, A.M. et al. A meta-analysis of the effect of different neuroprotective drugs in management of patients with traumatic brain injury. Neurosurg Rev 41, 427–438 (2018). https://doi.org/10.1007/s10143-016-0775-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-016-0775-y