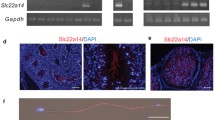
Abstract
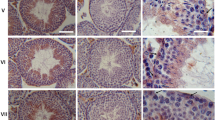
Spermatogenesis starts within the seminiferous tubules of the testis by mitotic division of spermatogonia that produces spermatocytes. Meiotic division of these spermatocytes produces haploid spermatids that differentiate into spermatozoa. In this study, we examined the expression of ENaC and CFTR (a Cl− channel) in rat testicular sections using confocal microscopic immunofluorescence. The structural integrity of the seminiferous tubule sections was verified by precise phalloidin staining of the actin fibers located abundantly at both basal and adluminal tight junctions. The acrosome forming regions in the round spermatids were stained using an FITC coupled lectin (wheat germ agglutinin). In all phases of the germ cells (spermatogonia, spermatocytes, and spermatids) ENaC was localized in cytoplasmic pools. Prior to spermiation, ENaC immunofluorescence appeared along the tails of the spermatids. In spermatozoa isolated from the epididymis, ENaC was localized at the acrosome and a central region of the sperm flagellum. The mature sperm are transcriptionally silent. Hence, we suggest that ENaC subunits in cytoplasmic pools in germ cells serve as the source of ENaC subunits located along the tail of spermatozoa. The locations of ENaC is compatible with a possible role in the acrosomal reaction and sperm mobility. In contrast to ENaC, CFTR immunofluorescence was most strongly observed specifically within the Sertoli cell nuclei. Based on the nuclear localization of CFTR we suggest that, in addition to its role as an ion channel, CFTR may have an independent role in gene regulation within the nuclei.











Similar content being viewed by others
References
Alves MG, Sá R, Jesus TT, Sousa M, Oliveira PF (2015) CFTR regulation of aquaporin-mediated water transport: a target in male fertility. Curr Drug Targets 16:993–1006
Baker MA (2011) The’omics revolution and our understanding of sperm cell biology. Asian J Androl 13:6–10. https://doi.org/10.1038/aja.2010.62
Beltrán C, Treviño CL, Mata-Martínez E, Chávez JC, Sánchez-Cárdenas C, Baker M, Darszon A (2016) Role of ion channels in the sperm acrosome reaction. In: Advances in anatomy, embryology, and cell biology. Springer, New York, pp 35–69
Bonny O, Hummler E (2000) Dysfunction of epithelial sodium transport: from human to mouse. Kidney Int 57:1313–1318. (https://doi.org/10.1046/j.1523-1755.2000.00968.x)
Boockfor FR, Morris RA, DeSimone DC, Hunt DM, Walsh KB (1998) Sertoli cell expression of the cystic fibrosis transmembrane conductance regulator. Am J Physiol 274:C922–C930
Chalmel F, Com E, Lavigne R, Hernio N, Teixeira-Gomes A-P, Dacheux J-L, Pineau C (2014) An integrative omics strategy to assess the germ cell secretome and to decipher sertoli-germ cell crosstalk in the Mammalian testis. PLoS ONE 9:e104418. https://doi.org/10.1371/journal.pone.0104418
Chan HC (2013) CFTR and male fertility—impact beyond cystic fibrosis. Spermatogenesis 3:e26228. https://doi.org/10.4161/spmg.26228
Chang SS, Grunder S, Hanukoglu A, Rösler A, Mathew PM, Hanukoglu I, Schild L, Lu Y, Shimkets RA, Nelson-Williams C et al (1996) Mutations in subunits of the epithelial sodium channel cause salt wasting with hyperkalaemic acidosis, pseudohypoaldosteronism type 1. Nat Genet 12:248–253. https://doi.org/10.1038/ng0396-248
Chen H, Ruan YC, Xu WM, Chen J, Chan HC (2012) Regulation of male fertility by CFTR and implications in male infertility. Hum Reprod Update 18:703–713. https://doi.org/10.1093/humupd/dms027
Chojnacka K, Zarzycka M, Mruk DD (2016) Biology of the sertoli cell in the fetal, pubertal, and adult mammalian testis. Results Probl Cell Differ 58:225–251. https://doi.org/10.1007/978-3-319-31973-5_9
Collawn JF, Lazrak A, Bebok Z, Matalon S (2012) The CFTR and ENaC debate: how important is ENaC in CF lung disease? Am J Physiol Lung Cell Mol Physiol 302:L1141–L1146. https://doi.org/10.1152/ajplung.00036.2012
Dimauro I, Pearson T, Caporossi D, Jackson MJ (2012) A simple protocol for the subcellular fractionation of skeletal muscle cells and tissue. BMC Res Notes 5:513. https://doi.org/10.1186/1756-0500-5-513
Dym M, Clermont Y (1970) Role of spermatogonia in the repair of the seminiferous epithelium following X-irradiation of the rat testis. Am J Anat 128:265–281. https://doi.org/10.1002/aja.1001280302
Edelheit O, Hanukoglu I, Gizewska M, Kandemir N, Tenenbaum-Rakover Y, Yurdakök M, Zajaczek S, Hanukoglu A (2005) Novel mutations in epithelial sodium channel (ENaC) subunit genes and phenotypic expression of multisystem pseudohypoaldosteronism. Clin Endocrinol 62:547–553. https://doi.org/10.1111/j.1365-2265.2005.02255.x
Edelheit O, Hanukoglu I, Dascal N, Hanukoglu A (2011) Identification of the roles of conserved charged residues in the extracellular domain of an epithelial sodium channel (ENaC) subunit by alanine mutagenesis. Am J Physiol Renal Physiol 300:F887–F897. https://doi.org/10.1152/ajprenal.00648.2010
Edelheit O, Ben-Shahar R, Dascal N, Hanukoglu A, Hanukoglu I (2014) Conserved charged residues at the surface and interface of epithelial sodium channel subunits—roles in cell surface expression and the sodium self-inhibition response. FEBS J 281:2097–2111. https://doi.org/10.1111/febs.12765
Enuka Y, Hanukoglu I, Edelheit O, Vaknine H, Hanukoglu A (2012) Epithelial sodium channels (ENaC) are uniformly distributed on motile cilia in the oviduct and the respiratory airways. Histochem Cell Biol 137:339–353. https://doi.org/10.1007/s00418-011-0904-1
Fok KL, Chen H, Ruan YC, Chan HC (2014) Novel regulators of spermatogenesis. Semin Cell Dev Biol 29:31–42. https://doi.org/10.1016/j.semcdb.2014.02.008
França LR, Hess RA, Dufour JM, Hofmann MC, Griswold MD (2016) The sertoli cell: one hundred fifty years of beauty and plasticity. Andrology 4:189–212. https://doi.org/10.1111/andr.12165
Giraldez T, Rojas P, Jou J, Flores C, Alvarez de la Rosa D (2012) The epithelial sodium channel δ-subunit: new notes for an old song. Am J Physiol Renal Physiol 303:F328-F338. https://doi.org/10.1152/ajprenal.00116.2012
Gur Y, Breitbart H (2006) Mammalian sperm translate nuclear-encoded proteins by mitochondrial-type ribosomes. Genes Dev 20:411–416. https://doi.org/10.1101/gad.367606
Hanukoglu I (2017) ASIC and ENaC type sodium channels: conformational states and the structures of the ion selectivity filters. FEBS J 284:525–545. https://doi.org/10.1111/febs.13840
Hanukoglu I, Hanukoglu A (2016) Epithelial sodium channel (ENaC) family: phylogeny, structure-function, tissue distribution, and associated inherited diseases. Gene 579:95–132. https://doi.org/10.1016/j.gene.2015.12.061
Hanukoglu A, Bistritzer T, Rakover Y, Mandelberg A (1994) Pseudohypoaldosteronism with increased sweat and saliva electrolyte values and frequent lower respiratory tract infections mimicking cystic fibrosis. J Pediatr 125:752–755. (https://doi.org/10.1016/S0022-3476(06)80176-9)
Hanukoglu A, Edelheit O, Shriki Y, Gizewska M, Dascal N, Hanukoglu I (2008) Renin-aldosterone response, urinary Na/K ratio and growth in pseudohypoaldosteronism patients with mutations in epithelial sodium channel (ENaC) subunit genes. J Steroid Biochem Mol Biol 111:268–274. https://doi.org/10.1016/j.jsbmb.2008.06.013
Hanukoglu I, Boggula VR, Vaknine H, Sharma S, Kleyman T, Hanukoglu A (2017) Expression of epithelial sodium channel (ENaC) and CFTR in the human epidermis and epidermal appendages. Histochem Cell Biol 147:733–748. https://doi.org/10.1007/s00418-016-1535-3
Hernández-González EO, Sosnik J, Edwards J, Acevedo JJ, Mendoza-Lujambio I, López-González I, Demarco I, Wertheimer E, Darszon A, Visconti PE (2006) Sodium and epithelial sodium channels participate in the regulation of the capacitation-associated hyperpolarization in mouse sperm. J Biol Chem 281:5623–5633. https://doi.org/10.1074/jbc.M508172200
Jiang L, Jin J, Wang S, Zhang F, Dai Y, Shi L, Zhang S (2017) CFTR gene mutations and polymorphism are associated with non-obstructive azoospermia: from case-control study. Gene 626:282–289. https://doi.org/10.1016/j.gene.2017.04.044
Kong X-B, Ma H-G, Li H-G, Xiong C-L (2009) Blockade of epithelial sodium channels improves sperm motility in asthenospermia patients. Int J Androl 32:330–336. https://doi.org/10.1111/j.1365-2605.2008.00864.x
Kreda SM, Mall M, Mengos A, Rochelle L, Yankaskas J, Riordan JR, Boucher RC (2005) Characterization of wild-type and deltaF508 cystic fibrosis transmembrane regulator in human respiratory epithelia. Mol Biol Cell 16:2154–2167. https://doi.org/10.1091/mbc.E04-11-1010
Leblond CP, Clermont Y (1952) Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann N Y Acad Sci 55:548–573
Li C, Naren AP (2010) CFTR chloride channel in the apical compartments: spatiotemporal coupling to its interacting partners. Integr Biol 2:161. https://doi.org/10.1039/b924455g
Li N, Tang EI, Cheng CY (2016) Regulation of blood–testis barrier by actin binding proteins and protein kinases. Reproduction 151:R29–R41. https://doi.org/10.1530/REP-15-0463
Lishko PV, Kirichok Y, Ren D, Navarro B, Chung J-J, Clapham DE (2012) The control of male fertility by spermatozoan ion channels. Annu Rev Physiol 74:453–475. https://doi.org/10.1146/annurev-physiol-020911-153258
Mruk DD, Cheng CY (2015) The Mammalian blood-testis barrier: its biology and regulation. Endocr Rev 36:564–591. https://doi.org/10.1210/er.2014-1101
O’Donnell L, Stanton PG, Bartles JR, Robertson DM (2000) Sertoli cell ectoplasmic specializations in the seminiferous epithelium of the testosterone-suppressed adult rat. Biol Reprod 63:99–108
O’Donnell L, Nicholls PK, O’Bryan MK, McLachlan RI, Stanton PG (2011) Spermiation Spermatogenesis 1:14–35. https://doi.org/10.4161/spmg.1.1.14525
Painter RG, Valentine VG, Lanson NA, Leidal K, Zhang Q, Lombard G, Thompson C, Viswanathan A, Nauseef WM, Wang G et al (2006) CFTR expression in human neutrophils and the phagolysosomal chlorination defect in cystic fibrosis. Biochemistry 45:10260–10269. https://doi.org/10.1021/bi060490t
Potter SJ, DeFalco T (2017) Role of the testis interstitial compartment in spermatogonial stem cell function. Reproduction 153:R151–R162. https://doi.org/10.1530/REP-16-0588
Rato L, Socorro S, Cavaco JEB, Oliveira PF (2010) Tubular fluid secretion in the seminiferous epithelium: ion transporters and aquaporins in Sertoli cells. J Membr Biol 236:215–224. https://doi.org/10.1007/s00232-010-9294-x
Rato L, Alves MG, Socorro S, Duarte AI, Cavaco JE, Oliveira PF (2012) Metabolic regulation is important for spermatogenesis. Nat Rev Urol 9:330–338. https://doi.org/10.1038/nrurol.2012.77
Sorio C, Buffelli M, Angiari C, Ettorre M, Johansson J, Vezzalini M, Viviani L, Ricciardi M, Verzè G, Assael BM et al (2011) Defective CFTR expression and function are detectable in blood monocytes: development of a new blood test for cystic fibrosis. PLoS ONE 6:e22212. https://doi.org/10.1371/journal.pone.0022212
Stanton PG, Foo CFH, Rainczuk A, Stephens AN, Condina M, O’Donnell L, Weidner W, Ishikawa T, Cruickshanks L, Smith LB et al (2016) Mapping the testicular interstitial fluid proteome from normal rats. Proteomics 16:2391–2402. https://doi.org/10.1002/pmic.201600107
Waldmann R, Champigny G, Bassilana F, Voilley N, Lazdunski M (1995) Molecular cloning and functional expression of a novel amiloride-sensitive Na + channel. J Biol Chem 270:27411–27414
Zhou Q, Clarke L, Nie R, Carnes K, Lai L-W, Lien Y-HH, Verkman A, Lubahn D, Fisher JS, Katzenellenbogen BS et al. (2001) Estrogen action and male fertility: roles of the sodium/hydrogen exchanger-3 and fluid reabsorption in reproductive tract function. Proc Natl Acad Sci USA 98:14132–14137. https://doi.org/10.1073/pnas.241245898
Acknowledgements
This research was funded in part by a grant from the United States-Israel Bi-national Science Foundation (BSF Grant Number 2011370).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
A cross-section of testis was reacted with DAPI and secondary antisera alone. The blue-colored DAPI staining of cell nuclei was visible but red immunofluorescence did not appear in such slides. Scale bar 20 μm (PNG 1496 KB)
Supplementary Fig. 2
Confocal microscopic imaging of rat sperm reacted with: (a) DAPI (blue); (b) anti-α-tubulin antisera (green); (c) goat anti-rabbit Alexa Fluor 555 IgG (secondary antibody used for ENaC antisera); and (d) merged image of (a), (b), and (c). Note that the sperm nuclei stained with DAPI have the typical hooked shape for rat sperm, and the anti-tubulin shows very specific staining along the whole length of the sperm tails. The secondary antibody used for ENaC immunofluorescence did not stain sperm (c). Scale bar 10 μm (PNG 1229 KB)
Supplementary Fig. 3
A tile-scan overview image showing the expression of CFTR in many seminiferous tubules of the rat testis. The pattern of CFTR immunofluorescence observed in all the seminiferous tubules was in the consistency with previous CFTR images, confirming that CFTR is uniformly localized in the nuclei of the Sertoli cells. Scale bar 100 μm (PNG 124 KB)
Rights and permissions
About this article
Cite this article
Sharma, S., Hanukoglu, A. & Hanukoglu, I. Localization of epithelial sodium channel (ENaC) and CFTR in the germinal epithelium of the testis, Sertoli cells, and spermatozoa. J Mol Hist 49, 195–208 (2018). https://doi.org/10.1007/s10735-018-9759-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-018-9759-2